Following on from my article about dual amylin calcitonin receptors agonists (DACRA) recently Abbvie and Zealand Pharma released topline data from their DACRA medications. I felt it was worth a short article exploring these results and what it means for both companies' ambitions for obesity medicine going forward. We also had topline data for a brand-new player in the obesity space. Regeneron and Hansoh Pharmaceutical released data on their dual GIP/GLP1 Olatorepatide, the results of which seem almost too good to be true. But first the DACRAs!
Petrelintide phase 2 obesity data
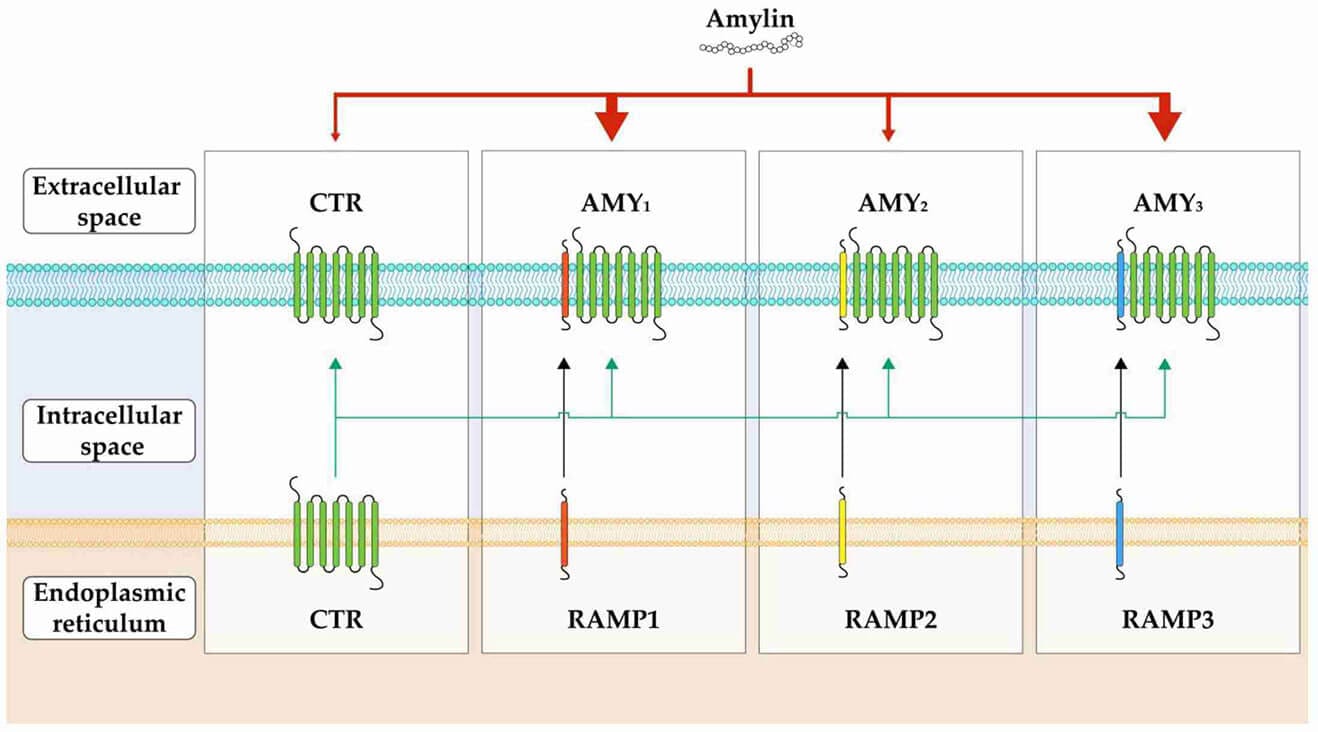
Petrelintide was developed by Zealand Pharma, while the company likes to refer to it in press releases as an ‘amylin analog,’ the reality is that it is a DACRA and was designed that way. From Zealand’s published data and, ironically, published Eloralintide data, I was able to find binding affinities for it. It has a 5-fold affinity for Amylin 1 over calcitonin and then essentially has balanced Amylin 3 and Calcitonin activity and has similar affinity as cagrilintide for those two receptors, hence it is a DACRA. For comparison, Eloralintide has a 12x affinity for Amylin 1 versus calcitonin and has minimal activity at Amylin 3 and calcitonin, hence why it is called a selective amylin receptor agonist (SARA)

Thanks, Lilly, for doing your diligence on your competition
All that out of the way, Zealand did themselves no favors by claiming that they expected best in class tolerability and weight loss. Some of us may remember Novo Nordisk claiming CagriSema would hit 25% weight loss and we’ve all seen what happened when they missed that target. Zealand claimed to expect 15-20% weight loss.

Probably should not overpromise things on a slide deck that is public record
In reality it was 10.7% weight loss. And Zealand described it thusly, “Double-digit body weight loss sustained through week 42, suggesting the potential for continued weight loss with longer treatment” Now that’s definitely nowhere near 15% weight loss, and the phrasing is weird. Zealand also didn’t realize what the doses were but there was almost no variability in weight loss between the dose arms, suggesting either they didn’t test high enough of a dose, or assuming one of these is a high dose, there’s a ceiling being reached for weight loss.
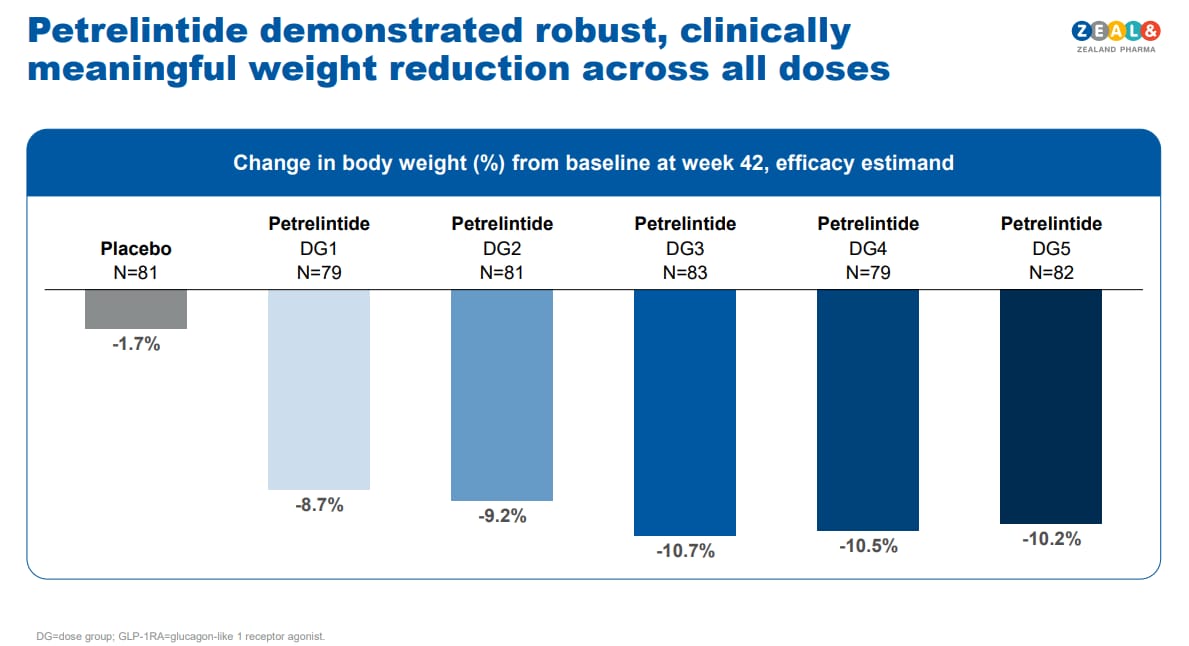
The lack of differences in weight loss between the dose groups is unusual
Moreover, they note their trial was 53% female and according to Zealand women lost 6% more than men, which doing some back of the napkin math means women were probably losing about 13.5% of their body weight versus 7.5% for men. Still not 15 percent and a really poor showing for men.
Also, can I take a moment to call out Eli Lilly as the Eloralintide trial was 77.6% female (but also even presuming the 6% difference in weight loss in men vs women with Eloralintide, it’s still causing significantly more weight loss than Petrelintide)
On the positive side, the side effect profile was as promised, essentially best in class, minimal nausea, no vomiting, minimal diarrhea and constipation and nearly 70% of the patients reporting NO gastrointestinal adverse events “at any time during the trial.” That is a fairly remarkable data point, especially because we’re just so used to GI side effects these days, it’s unfortunate that the weight loss wasn’t better.
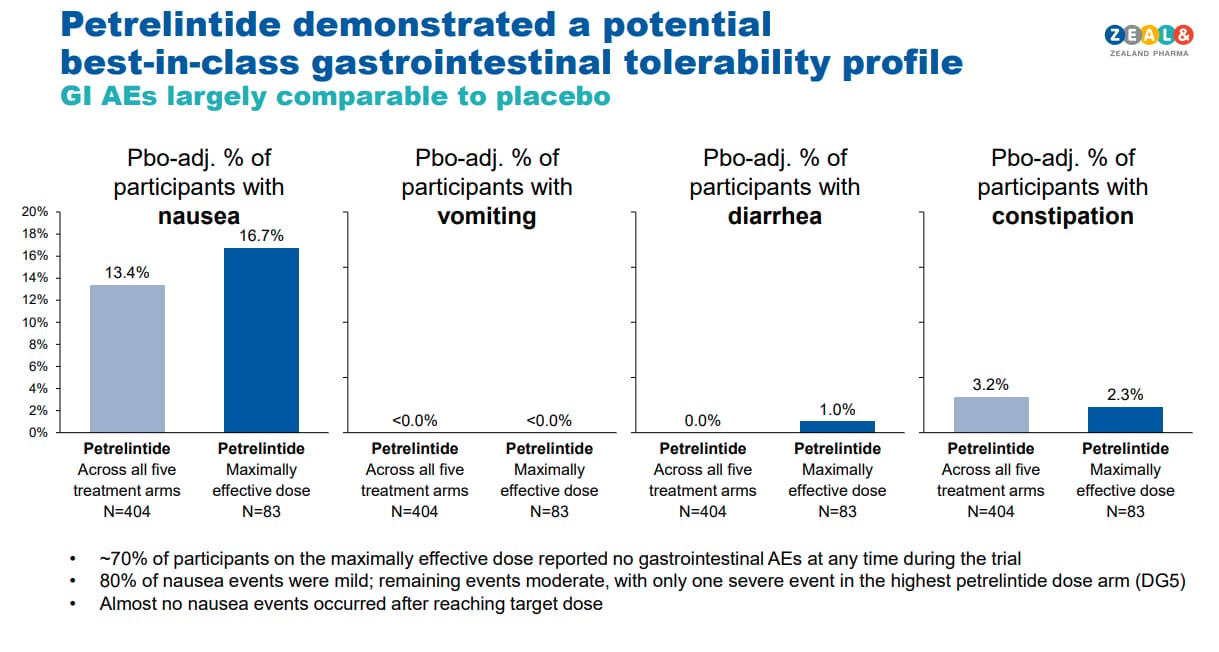
More of this please! Incredible side effect profile
I’m eager to see the weight loss curve for this drug, something Zealand didn’t provide but 10.7% weight loss at 42 weeks suggests a similar weight loss as Cagrilintide if one looks at the REDEFINE-1 data as a rough comparison. All that to say, I’d love to see this on the market given the lack of side effects, but it may be better for folks that don’t need a lot of weight to lose such as patients with a BMI under 35 or even aesthetic weight loss if managed cautiously. Zealand & Roche do plan to combine it with CT-388 which is a dual GIP/GLP-1 medication that ironically suffers from a high amount of GI side effects at higher doses. Maybe high dose Petrelintide plus low dose CT-388 gets you into the 20% weight loss range and a more competitive place in the market?
ABBV-295 phase 1 obesity data
Moving on from there we have Abbvie, a relatively new player in the obesity medicine space, and their partnership with Gubra with their DACRA, now labeled ABBV-295. This particular molecule I have very limited data, it has an 11-day half-life, but I have no data on binding affinities or anything like that. This was a phase 1 multi-ascending dose trial testing weekly, every other week and “monthly after week 5” dosing.

Right out of the gate, the extended dosing arms especially the “monthly after week 5” don’t show as much weight loss as weekly dosing, but this study was short, only 13 weeks, so ask again later if that’s a viable dosing strategy. With cohort 5a there was nearly 10% weight loss in 12 weeks which is on par with Eloralintide phase 2 data and this is doubly impressive because the cohort was 88% men!
However, there was only a generic line about side effects being ‘mostly mild.’ So, take that how you want? Good weight loss but ?????? side effects. I’m not sure how to describe this as Abbvie management didn’t seem pleased despite matching Eloralintide weight loss curves. Abbvie has indicated they’ll continue development of this but perhaps the molecule wasn’t as well tolerated as they expected? Check back in a year or two to see if anything becomes of this.
Olatorepatide phase 3 obesity data
The final trial data was from another newcomer to the obesity medicine space, that being Regeneron. They partnered with Hansoh Pharma in China to develop an asset called olatorepatide. This is a dual GIP/GLP1 agonist that has just completed its first phase 3 trial in China.
The data here is perhaps even MORE sparse than the others. I was able to locate the actual amino acid sequence of the drug and to my eye it appears to be heavy on the GIP and light on the GLP-1 in terms of what Hansoh was aiming for in development. However, I don’t have any binding affinity to be able to confirm this.
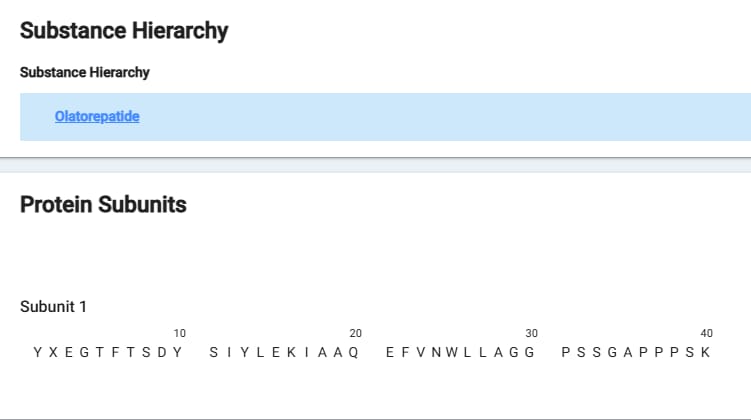
This sequence is very similar to other GLP-1/GIP dual agonists but an unusual fatty acid linker at the end of the sequence. Usually this is found mid-sequence, this may explain the tolerability profile
Otherwise, this trial was a phase 3 obesity trial, conducted in China with 604 patients over 48 weeks. They tested doses of olatorepatide in 5mg, 10mg and 15mg doses (sound familiar, Tirzepatide users?) Mean weight loss was 19% at 48 weeks and that 97% of patients had >5% weight loss. They do not highlight dose specific weight loss; they don’t provide baseline BMI or any other characteristics. For a dual GIP/GLP-1 drug that all seems reasonable, but the next part is what strains credulity to me, they claim “The average incidence of nausea was below 10%, and the average incidence of vomiting below 5%.”
That’s well below any GLP-1 based medication that is currently available. I found some data from phase 1 and phase 2 trials that seem to show very good tolerability, but that level of tolerability would in a phase 3 trial would be game changing, even if it’s late to the market.

Phase 1 side effects were extremely low despite rapid titration, with slow phase 3 titration is it possible the side effects are as low as reported?
The only thing that I know of that approaches that level of tolerability is Macupatide which is Eli Lilly’s mono-agonist GIP drug in development. Now Regeneron is planning a full global phase 3 program for this drug, so we’ll rapidly find out if these low rates of side effects were a fluke or reality. They also plan to run phase 3 trials of olatorepatide and their PCSK9i cholesterol drug Praluent (alirocumab) on the theory that a 40-50% reduction in cholesterol plus a large weight loss is probably beneficial to cardiovascular outcomes. Well duh, of course it would.
Anyways, consider me skeptical on the side effect profile of olatorepatide until proven otherwise. Do what Zealand did, come with receipts please instead of just claiming it in your press release! It is encouraging to see companies pivoting towards tolerability instead of just raw weight loss, we need more of that as side effects are still a very common reason for patients to stop any of these drugs. Definitely a trend to keep an eye on going into 2027.
Stay tuned! Next article will be on the anti-inflammatory benefits of GLP-1 medications
References: