As my last article alluded to, 2026 is a critical year for Novo Nordisk when it comes to one drug in particular. CagriSema. I said it would make or break their year and so far, it seems to be the latter. They recently reported topline results for REIMAGINE-2 which was a trial of CagriSema vs Semaglutide in type 2 diabetes with primary outcomes of weight loss and A1c change. The weight loss number was similar to past results, about 14% weight loss, but the A1c change landed with a dull thud. In this trial 2.4mg of semaglutide reduced A1c by 1.76% and CagriSema 2.4mg reduced A1c by 1.91%. Novo did claim this was a statistically significant difference. However, I am suspicious of that claim given prior A1c reductions obtained with semaglutide alone. As always, the full published data will tell the tale
Even more auspiciously this trial included a Cagrilintide arm alone but did not report the weight changes or A1c changes for that particular arm of the trial…which is certainly a choice sure to inspire confidence. The only other phase 3 diabetic trial of CagriSema showed a 2.1% reduction in A1c, but even that is in the realm of what semaglutide alone can manage. Then on their February 4th Q4 2025 earnings call, Novo snuck in a mention of REIMAGINE-3 and noted that CagriSema added to basal insulin reduced A1c 2.33% in only 40 weeks. This deepened the mystery for me that something is off with this drug and diabetes.
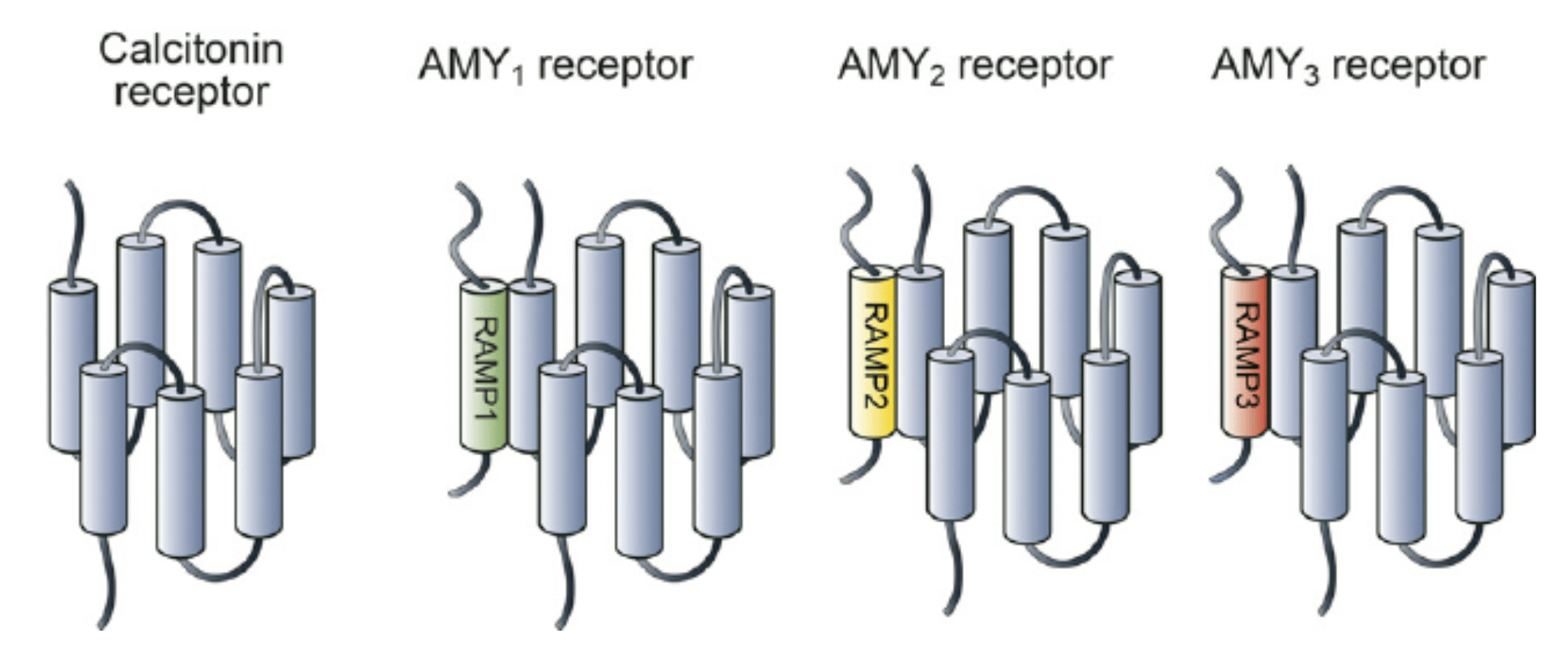
Meanwhile, over at Eli Lilly they only have one Amylin drug in development, Eloralintide, and as your author can attest to, Lilly was very proud to discuss at Obesity Week 2025 that, unlike Cagrilintide, their drug was a selective Amylin receptor agonist(SARA) only and had very minimal activity at calcitonin receptors, which is an oddly specific thing to tout. The rest of the industry has mostly focused on developing DACRA (dual amylin, calcitonin receptor agonist) and I found it odd for Lilly to basically ignore one receptor while celebrating the other. They have also quickly advanced Eloralintide into phase 3 trials for type 2 diabetics, both alone and in combination with other drugs.
Novo has been slow to do the same with Cagrilintide by itself, only recently starting phase 3 Type 2 diabetes trials despite having trials of Cagrilintide extending back to 2019 and as noted, not even releasing the data on Cagrilintide data from REIMAGINE-2. In fact, the phase 2 trials of Cagrilintide were completed in 2021 but not published until 2023. The results were a bit underwhelming.

Pardon the poor quality, focus on the fasting insulin drop, but not a drop in glucose, unlike liraglutide which decreases glucose and insulin, and no drop in A1c.

Minimal change in HOMA-IR suggesting no change in insulin sensitivity, and HOMA-B reflecting the decrease in insulin secretion
All this introductory backstory to get to why I’m writing this article: CagriSema is a pharmacological contradiction. It does allow synergistic weight loss, however I believe the calcitonin agonism aspect of this particular DACRA is actively antagonistic to the actions of GLP-1, especially in diabetic patients. I also believe that some of this relates to differences in animal models of calcitonin versus what actually happens in humans. This wouldn’t be something new, many times in pharmacology, an effect is seen in animal models that doesn’t translate to humans at all.
So that’s my hypothesis, but where’s the evidence? And what does this mean for Novo and multiple other DACRA drugs in development?
To secret insulin or to not, that is the question
One goal I have for this blog in 2026 was to write articles on how GLP-1 and how Amylin works in the body since they’re so commonly discussed and share so much spotlight. Those articles will be forthcoming, but for now we will focus on one mechanism of how GLP-1 in particular works.
Insulin secretion.
GLP-1 is what we refer to as an insulin secretagogue, and this is one of the reasons it works so well as a drug for diabetes. It initially promotes the release of insulin which lowers blood glucose, but only back to normoglycemia. With time, insulin production actually drops as glucose levels revert towards normal. Lower glucose and insulin levels can then help with some of the weight loss as well. GLP-1 also tends to lower secretion of glucagon as well which can also further reduce blood glucose. Now, it has an effect elsewhere in the body as well but for today, the important part is insulin secretion for diabetics. You can see this illustrated extremely well from the orforglipron phase 3 trials in diabetics:


Amylin on the other hand is NOT an insulin secretagogue. Rather it is co-secreted with insulin at a ratio of about 1 unit of amylin to every 100 units of insulin. For this article its primary purposes are threefold: 1.) slowing gastric emptying and delaying nutrient absorption, especially glucose which delays large spikes in blood sugar, this in turn 2.) indirectly decreases glucagon production in the liver which also helps keep glucose under control, finally 3.) It works in the brain to act as a “stop” eating or meal termination signal, increasing satiety.
Taken together amylin and GLP-1 seem like they would pair fairly well on first blush in terms of a weight loss drug. Maybe not ideal for diabetes, as a mechanism to increase insulin sensitivity isn’t present outside of weight loss but it should get the job done. I’m skipping over lots of nuance here, but one can easily see why Novo Nordisk and indeed other companies have looked to Amylin and have openly discussed combining it with GLP-1 which is how CagriSema came into existence.
But I’m leaving out a crucial part: calcitonin. Amylin gets all the press coverage and calcitonin is sitting in the back of the room completely unnoticed and this is where I think Novo and others are missing something crucial. I’m going to quote directly from this paper: THERAPY OF ENDOCRINE DISEASE: Amylin and calcitonin – physiology and pharmacology | European Journal of Endocrinology | Oxford Academic
“…several studies point to an acute reduction in insulin secretory responses to oral glucose or a meal after administration of salmon calcitonin. This observation may relate to the delayed gastric emptying rate, but basal insulin and the insulinotropic response to intravenous glucose are also affected by salmon calcitonin. Thus, salmon calcitonin may exert an effect on insulin secretion via other mechanisms”
The link to those other studies is in the references at the end of this article for full transparency's sake. Caveats that salmon calcitonin isn’t human calcitonin however, it’s still activating the receptor which is what we’re looking for.
For a diabetic patient, a blunted insulin response is not what you’d like to see, however, I think when looking at early data from Cagrilintide that’s exactly what I can see both in non-diabetics and in diabetics across two different trials, and as I opened with in the beginning that is antagonistic to what GLP-1 is doing.

Hints back in phase 2 that CagriSema was only better at weight loss, but not A1c.
Just look at the green trend line for A1c reduction with cagrilintide alone, it's there, but not what we usually see with other incretins. Notice the immediate drop with CagriSema and Semaglutide? I think that’s essentially all GLP-1. Combined with semaglutide it looks more like other incretins. With cagrilintide alone the glucose and insulin reductions are not nearly what you’d think to see given the amount of weight loss achieved. Moreover, digging into the supplemental appendix for this trial, once again finds minimal changes in insulin and glucagon.
I believe this is what’s really limiting CagriSema as a diabetic drug(along with side effects, more on that in a moment) and what makes tirzepatide remarkable in comparison is not just the weight reductions, but the dramatic changes in insulin sensitivity, glucose disposal/uptake and reductions in fasting insulin that are then a result of those two things. Meanwhile reducing insulin secretion while keeping insulin sensitivity the same just doesn’t confer the same benefit. As I noted in the opening, while writing this article Novo Nordisk very quietly slipped out topline data for REIMAGINE 3, which was CagriSema added to basal insulin for diabetics and the A1c reduction was the best seen yet, 2.33% reduction! Not to be sarcastic but adding insulin back to the equation helped!
Even when looking at the full phase 3 data from the REDEFINE 1 & 2 trials Novo is extremely shy with showing changes in fasting insulin levels without actually diving into the appendix. For the REDEFINE 2, a trial looking at diabetes, one cannot find changes in HOMA-B or HOMA-IR(the way we measure insulin sensitivity) In REDEFINE-1 they list a change in HOMA-IR for CagriSema but not for any of the other comparator arms so it’s difficult to draw any conclusions from that lone number.
Unfortunate synergies and unbalanced agony agonism
There’s another reason that calcitonin is a liability in my mind.
Side effects.
In this I can pull from actual approved drugs to prove my point. We use salmon calcitonin to help treat various diseases, most commonly osteoporosis as it helps to regrow/preserve bone mass.
What are some of the more common side effects?
Nausea, vomiting, diarrhea, fatigue, headache, dizziness. These are essentially the same side effects as GLP-1 and when combined the two have unfortunately synergistic effects as seen in the phase 3 data, especially for nausea and vomiting.
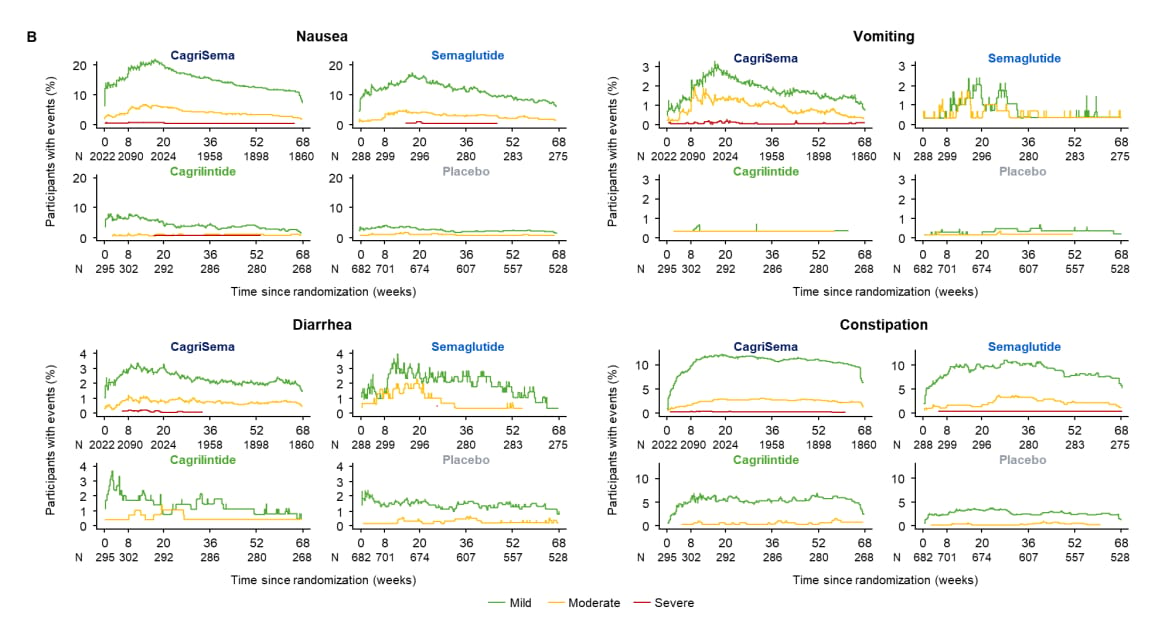
Noticeably more nausea and vomiting with CagriSema
These effects of calcitonin weren’t unknown and in fact probably could have easily been predicted but somewhere along the way that knowledge was either lost or brushed aside? I’m not sure. What I do suspect is that they failed to translate pre-clinical models to humans.
I mentioned insulin sensitivity multiple times now as something we don’t see with cagrilintide. In fact, Novo only has one that graphic depicting HOMA-IR from early on in development and the results weren’t impressive. However, a quick search on PubMed will yield multiple trials in pre-clinical models showing various calcitonin agonists, DACRAs etc improving insulin sensitivity in rodent models and yet in humans we continue to see the opposite as shown earlier. I can even go so far as to point out a molecule call KBP-042 which was a DACRA, did not improve A1c in diabetics according to the company developing the molecule.
In fact, the only drug that we have decent data on so far that reduces glucose, A1c and insulin in a manner consistent with GLP-1 agonists is eloralintide and as noted it has minimal calcitonin activity compared to Cagrilintide. That suggests the preclinical data with DACRAs was perhaps amylin driven and not calcitonin.
Pop quiz with these two images, which one is a GLP-1 agonist and which one is an amylin agonist?

Is this a GLP-1 or a SARA?
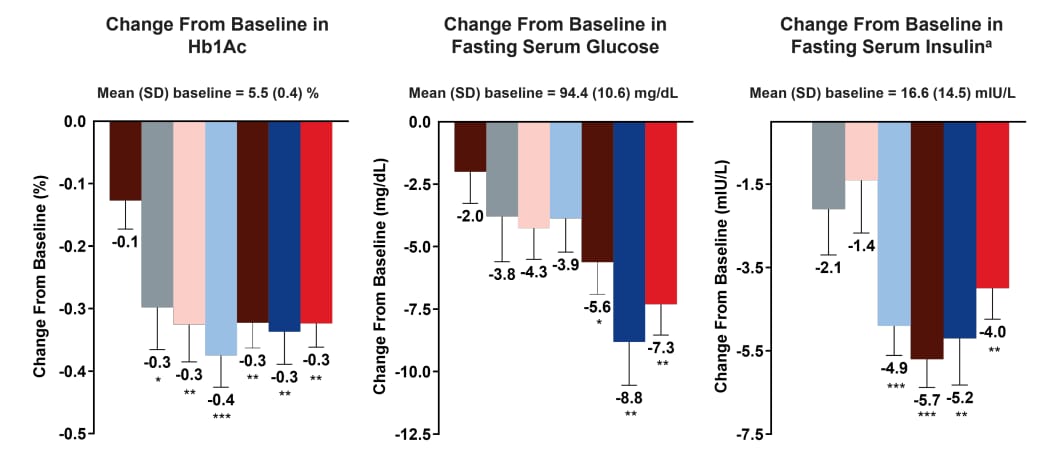
What about this?
To answer the pop quiz, the first one is data from orforglipron and the second one is eloralintide. They are exceedingly similar, right on down to the insulin changes, about 20-35% reduction in serum insulin levels. I believe this is why Lilly can have a trial with eloralintide in diabetics as a monotherapy, but we won’t find out that data until later this year.
The last reason is receptor balance and binding affinity. Cagrilintide is like a shotgun approach to Amylin and calcitonin receptors, as are most of the other DACRAs in development. What I mean in very simple terms is that it activates those receptors broadly. Whereas Eloralintide is basically a laser beam for the Amylin 1 receptor. This receptor balance matters as we’ve seen in the GLP-1 family of medication. Tirzepatide is famously an unbalanced dual agonist that has more binding affinity for GIP than GLP-1 and it’s thought this helps to contribute to its tolerability as other GIP/GLP-1 dual agonists have often favored GLP-1 activity over GIP, which leads to an increase in side effects instead. I think the same is true here, more amylin affinity equals lower side effects, and more calcitonin affinity is the opposite, less tolerability.
AstraZeneca has a SARA in development that is unbalanced towards the Amylin 3 receptor, in aversion testing in rats, there was significantly less sweetness aversion when compared to CagriSema or Cagrilintide alone.
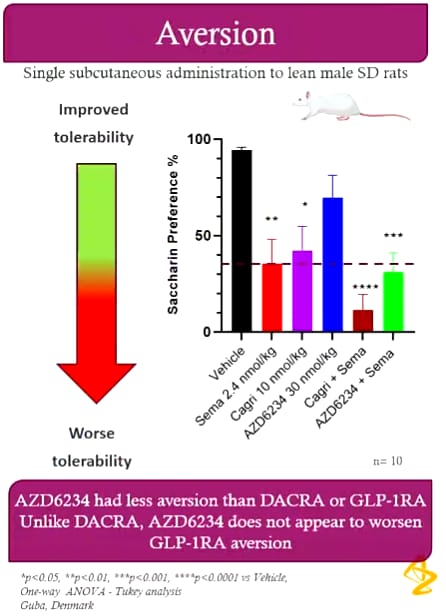
In rats, but important to consider for humans as well
So how did this happen?
I’ve mentioned a couple possible reasons earlier on, but I’d also suggest a simple psychological one, sunk cost fallacy. That is, according to the Oxford dictionary, the phenomenon whereby an entity is reluctant to abandon a strategy or course of action because they have invested heavily in it, even when it is clear that abandonment would be more beneficial. Now I’m not saying Novo should abandon the drug, I do think it’ll have a place both for diabetes and especially obesity, but rather they thought they had the next best thing on their hands and, in poker terms they went all in on CagriSema, and when the results weren’t what they expected they doubled down again because they no other choice. They’d devoted so much time and effort into this development while at the same time the rest of their development pipeline faltered which was honestly just bad luck as far as I can tell.
This goes for the rest of the industry as well, the pharma industry is very much a copycat industry and so when Novo had success, everyone else followed suit to make their own DACRA. Only two companies didn’t play follow the leader, Lilly and AstraZeneca. One of these companies has shown great results so far with eloralintide, with the other company likely to show similar results come summer 2026. We’ll see what the rest of the DACRAs bring, but as a class I think we’ll see a shift to selective amylin receptor agonists simply because they work just as well with less side effects. I can also tentatively say they don’t create a liability for diabetic patients. My goal here wasn’t to drag Novo, but rather point out a deep pattern I noticed and hopefully now you’ll look for it too as we see more data from other DACRAs this year. I also think a deeper dive into amylin deserves its own article given everything I’ve written here.
Stay tuned I’ve got multiple articles in the works!
References:
THERAPY OF ENDOCRINE DISEASE: Amylin and calcitonin – physiology and pharmacology | European Journal of Endocrinology | Oxford AcademicDoes receptor balance matter? – Comparing the efficacies of the dual amylin and calcitonin receptor agonists cagrilintide and KBP-336 on metabolic parameters in preclinical models - ScienceDirect